Journal of Hepatology
Article in Press
Jean-Michel Pawlotsky
Received 28 February 2013; received in revised form 22 March 2013; accepted 27 March 2013. published online 08 April 2013.
Corrected Proof
Summary
Hepatitis C virus infection is a major health problem worldwide and no vaccine has yet been developed against this virus. In addition, currently approved pharmacotherapies achieve suboptimal cure rates and have side effects that result in non-compliance and premature treatment discontinuation. Significant research has been devoted to developing direct-acting antiviral agents that inhibit key viral functions. In particular, several novel drug candidates that inhibit the viral non-structural protein 5A (NS5A) have been demonstrated to possess high potency, pan-genotypic activity, and a high barrier to resistance. Clinical trials using combination therapies containing NS5A inhibitors have reported results that promise high cure rates and raise the possibility of developing interferon-free, all-oral regimens.
Abbreviations: HCV, hepatitis C virus, DAA, direct acting antiviral, NS, nonstructural, RdRp, RNA-dependent RNA polymerase, IFN, interferon, UTR, untranslated region, IRES, internal ribosome entry site, SVR, sustained virological response, RVR, rapid virologic response, cEVR, complete early virologic response
Keywords: Hepatitis C virus, NS5A inhibitor, Daclatasvir, Resistance
Introduction
Recent estimates indicate that there are more than 120–130million chronic hepatitis C virus (HCV) carriers worldwide [1], who are at risk of developing cirrhosis and/or hepatocellular carcinoma (primary liver cancer). As many as 4million persons are thought to be chronically infected in the US [2], 5–10million in Europe [2], 12million in India [2], and 1.2million in Japan (2004 figure) [3]. Most of these individuals are not aware of their infection. The incidence of acute infection in the US has declined from 7.4/100,000 in 1982 to 0.7/100,000 in recent years, primarily due to screening of blood in transfusion centres and improved safety of intravenous drug use [4]. It is estimated that approximately 150,000 new cases occur annually in the United States and in Western Europe, and about 350,000 in Japan. Only 25% of acute cases are symptomatic, but up to 80% of these acute cases progress to chronic infection and liver disease, and up to 20% of chronic infections progress to cirrhosis [2]. Every year, 4–5% of cirrhotic patients develop hepatocellular carcinoma [5]. Despite the decrease in HCV incidence, the number of patients with chronic HCV-related complications is increasing in those aging patients who have been infected for many years, and chronic hepatitis C infection will continue to be a significant cause of premature mortality, causing at least 200,000–300,000 deaths per year worldwide [4].
A number of direct-acting antiviral agents (DAAs) are under development for the treatment of chronic HCV infection. These agents block viral production by directly inhibiting one of several steps of the HCV lifecycle. As shown in Fig. 1, the genomic organization of HCV has been elucidated, and several viral proteins involved in the HCV lifecycle, such as the non-structural (NS) 3/4A serine protease, the NS5B RNA-dependent RNA polymerase (RdRp), and the NS5A protein, have been targeted for drug development [4]. Two NS3/4A protease inhibitors, telaprevir and boceprevir, which inhibit post-translational processing of the HCV polyprotein into individual non-structural proteins, have been approved by the US Food and Drug Administration, the European Medicines Agency, and several other regulatory agencies for the treatment of chronic HCV genotype 1 infection in combination with pegylated interferon (IFN)-α and ribavirin [6], [7].
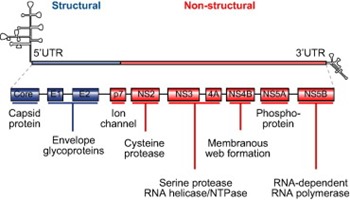 Fig,1. Structural organization of HCV RNA and viral proteins. NS, non-structural; UTR, untranslated region.
Fig,1. Structural organization of HCV RNA and viral proteins. NS, non-structural; UTR, untranslated region.
HCV structure and lifecycle, and physiological role of the NS5A protein
HCV is an enveloped virus with a single-stranded positive RNA genome of approximately 9.6kb. At the flanking ends of the genome are 2 highly conserved untranslated regions (UTRs). The 5′ UTR is highly structured and contains the internal ribosome entry site (IRES), which is important for the initiation of the cap-independent translation of the polyprotein [8]. The 3′ UTR consists of a short genotype-specific variable region, a tract consisting solely of pyrimidine residues (predominantly uridine) and a conserved 98-nucleotide sequence, known as X region, containing 3 stem-loops [9], [10]. The HCV open reading frame is situated between the two UTRs.
After entering the bloodstream, HCV binds to a receptor complex at the surface of its target cells, the hepatocytes. The envelope glycoproteins E1 and E2 are essential for target cell recognition, binding, and internalization [11]. The bound virus then undergoes clathrin-mediated endocytosis [12]. Acidification of the endocytosis vesicle frees the genomic RNA from the nucleocapsid for release into the cytoplasm. Along with host RNA molecules, the viral RNA migrates to the endoplasmic reticulum (ER). Binding of the 40S ribosomal subunit to the HCV IRES produces a stable pre-initiation complex that begins translation of the viral open reading frame to generate an approximately 3000 amino acid polyprotein. Following translation, the polyprotein is cleaved by both cellular and viral proteases to produce at least 10 viral proteins, including structural proteins (core, E1 and E2) and non-structural proteins (p7, NS2, NS3, NS4A, NS4B, NS5A, and NS5B) [13], [14].
Viral replication (i.e., the synthesis of new positive RNA genomes that may also serve as messenger RNAs for viral protein synthesis) is catalyzed by the viral RdRp, or NS5B protein. A negative-strand intermediate of replication is initially produced, which then serves as a template for the synthesis of numerous positive strands. The NS5A viral protein has been shown to play an important role in the regulation of replication. In addition, host cell proteins, such as cyclophilin A, act as necessary co-factors of HCV replication through their interactions with both NS5A and the RdRp in the replication complex [15], [16].
The non-structural NS5A protein bears pleiotropic functions, including roles in viral replication and assembly, and complex interactions with cellular functions. The latter include inhibition of apoptosis and promotion of tumorigenesis, both of which may play a role in the triggering of the hepatocarcinogenic process [17], [18], [19], [20]. The protein is comprised of approximately 447 amino acids and localizes to ER-derived membranes. It basally exists in phosphorylated (p56) and hyperphosphorylated (p58) forms that are implicated in different functions [21], [22], [23]. Its cytoplasmic moiety contains 3 domains, of which Domain I is the most conserved [24]. The mechanism by which NS5A regulates replication regardless of the HCV genotype is still unclear [25]. Considerable information has been gathered on its molecular interactions and role in the viral lifecycle. NS5A and the RdRp directly interact, both in vivo and in vitro [26]. In vitro, this interaction stimulates RdRp-catalyzed synthesis of the negative RNA strand [27]. It was shown that all 3 domains of NS5A bind to RNA [9]. The interactions of Domain I with the polypyrimidine tract of 3′ UTR suggest it may affect the efficiency of RNA replication by the RdRp; however, these results also suggested the binding of RdRp and NS5A to RNA are mutually exclusive. In addition, Domain II of NS5A interacts with cyclophilin A, a host cell protein required for replication, and this interaction is vital for RNA binding [28]. NS5A also plays a role in viral packaging and assembly. Domain III appears to be essential for this function [29], [30]. This may be due, at least in part, to NS5A recruiting apolipoprotein E, a component of the HCV production process [29], [31]. Indeed, inhibiting apolipoprotein E expression results in marked reduction of infectious particle production without affecting viral entry and replication [31].
NS5A inhibitor mechanism of action
Several viral proteins have generated interest as potential targets for specific inhibitory drugs. In addition to the two NS3/4A protease inhibitors already approved for clinical use, numerous other protease inhibitors are being developed as well as inhibitors of viral replication, including nucleoside/nucleotide analogue inhibitors of HCV RdRp, non-nucleoside inhibitors of RdRp, cyclophilin inhibitors, and NS5A inhibitors.
Because of its critical involvement in viral replication and assembly [32], NS5A has been identified as a target for viral inhibition, leading to development of therapeutic agents. In HCV replicon-containing cells, inhibition of NS5A, but not other HCV proteins, resulted in redistribution of NS5A from the ER to lipid droplets. NS5A-targeting agents did not cause similar alterations in the localization of other HCV-encoded proteins, and the transfer of NS5A to lipid droplets coincided with the onset of inhibition of replication [33]. Inhibition of NS5A at picomolar concentrations has been associated with significant reductions in HCV RNA levels in cell culture-based models, which makes these agents among the most potent antiviral molecules yet developed [34], [35], [36]. NS5A inhibitors have pan-genotypic activity, i.e., they suppress replication of all HCV genotypes, but their antiviral effectiveness against genotypes other than 1 may vary from one molecule to another [35]. Use of multiple DAAs including an NS5A inhibitor in replicon systems in cell culture has resulted in additive/synergistic inhibition of viral production and an increased barrier to resistance [37].
The exact mechanism of antiviral action of NS5A inhibitors is unknown. Available evidence suggests that they have multiple effects, which contribute to their potency [32]. One putative mechanism is the inhibition of hyperphosphorylation. Phosphorylation of NS5A seems required for viral production [38], but the relative roles of the phosphorylated and hyperphosphorylated forms are unclear, and conflicting results have been reported suggesting that reduced hyperphosphorylation may either enhance or reduce replication [21], [39]. It is thought that a tightly regulated control of phosphorylation vs. hyperphosphorylation is required for efficient viral function. It was also shown that NS5A acts in two different pathways in RNA replication, and one of them likely requires hyperphosphorylation [23]. However, other mechanisms may also play a role. For instance, NS5A inhibitors alter the subcellular localization of NS5A, which may cause faulty viral assembly [33], [40].
Resistance to NS5A inhibitors
HCV displays a large degree of genomic variability, resulting in its quasispecies distribution [41]. Variants that confer resistance to NS5A inhibitors pre-exist within HCV quasispecies populations in the absence of any previous exposure to these drugs. These variants generally replicate at low levels and are thus undetectable by currently available techniques. However, they can be selected if an NS5A inhibitor is administered and may be grown to high levels. Clinically significant resistance is usually associated with an escape pattern whereby viral replication returns to pretreatment levels and the dominant virus harbours amino acid substitutions that confer high levels of drug resistance without impairing fitness of the virus. Very high levels of the drug may be required to suppress highly resistant viruses, which may not be achievable without compromising safety [42].
At present, only genotype 1, the most prevalent HCV genotype, has been studied in detail for resistant variants. Table 1, adapted from Fridell et al. [43], describes the resistance profile of the NS5A inhibitor daclatasvir in genotype 1a and 1b replicons. The barrier to resistance is lower for genotype 1a than for genotype 1b. Substitutions at positions L31 and Y93 have the greatest ability to confer resistance to daclatasvir, and double mutations may increase the EC50 to a far greater extent (Table 1). These substitutions also confer resistance to other first-generation NS5A inhibitors. In addition, studies with daclatasvir have shown that double and triple inhibitor combinations in replicon systems can generate resistance pathways that differ from those observed during NS5A inhibitor monotherapy [37]. Agents without cross-resistance with NS5A inhibitors should thus be used in combination with this class of drugs.
Table 1. Resistance profile of daclatasvir in the in vitro genotype 1a and 1b replicon systems. Adapted from Fridell et al. [43].

NS5A inhibitors undergoing clinical trials
Although no NS5A inhibitor has yet been approved for therapeutic use, these agents are viewed with optimism due to their favourable characteristics, including the requirement for low dosing to inhibit HCV replication; pan-genotypic activity; once-daily dosing; resistance profiles that do not overlap with those of other DAAs in development; and successful suppression of HCV replication with an acceptable safety profile in early clinical trials [34].
Daclatasvir (BMS790052)
Daclatasvir is an oral, once-daily, highly selective NS5A inhibitor with broad coverage of HCV genotypes in vitro developed by Bristol-Myers Squibb. Daclatasvir currently is in Phase III clinical trials. Its inhibitory target maps to Domain I, and it has been shown to block hyperphosphorylation of NS5A [23], as well as alter the subcellular localization of the viral protein [33], [40]. Daclatasvir has an EC50 of 50pM against genotype 1a, 9pM against genotype 1b, and 28pM against genotype 2a [35]. Daclatasvir has been tested in Phase II clinical trials in combination with pegylated IFN-α and ribavirin; in quadruple combination with asunaprevir, an NS3/4A protease inhibitor, and pegylated IFN-α/ribavirin; and with asunaprevir, the nucleotide analogue sofosbuvir and the non-nucleoside inhibitor of HCV RdRp BMS-791325 in IFN-free regimens.
In a randomized, parallel-group, double-blind, placebo-controlled, dose-finding Phase IIa trial of treatment-naïve patients infected with HCV genotype 1, 5 of 12 patients who received 3mg daclatasvir with pegylated IFN-α and ribavirin for 48weeks achieved extended rapid virologic response (eRVR), compared with 10 of 12 who received 10mg daclatasvir, 9 of 12 who received 60mg daclatasvir, and 1 of 12 who received placebo. Adverse events and discontinuations as a result of adverse events occurred with similar frequency across treatment groups [44]. In another Phase IIa trial in genotype 1-infected patients who were non-responders to a prior course of pegylated IFN-α and ribavirin, all 10 patients who received quadruple therapy with daclatasvir, asunaprevir, and pegylated IFN-α/ribavirin showed a sustained virologic response (SVR) after 12weeks, as opposed to 4 of 11 who received daclatasvir and asunaprevir only [45]. A higher incidence of viral breakthrough due to resistance was observed in genotype 1a patients who were given only the 2 DAAs without pegylated IFN-α/ribavirin vs. genotype 1b patients receiving the same treatment regimen, as a result of the lower barrier to resistance in genotype 1a [45]. Among patients who experienced virologic failure, the most common variants harboured Y93H and L31M, two substitutions well known for conferring resistance to daclatasvir.
In a 24-week dual-oral Phase II trial with daclatasvir and asunaprevir in genotype 1b-infected patients, 90.5% of null responders and 63.6% of patients ineligible for or intolerant to pegylated IFN-α/ribavirin achieved SVR 24weeks after the end of treatment (SVR24) [46]. Interestingly, many patients in this study with pre-existing resistance-associated NS5A polymorphisms were cured of their chronic HCV infection.
In a Phase IIb study with daclatasvir, pegylated IFN-α, and ribavirin, 100% of genotype 4-infected patients achieved SVR at 12weeks post-treatment (SVR12) [47]. A combination of daclatasvir and sofosbuvir (formerly GS-7977), a nucleotide analogue inhibitor of HCV RdRp developed by Gilead Sciences, given for 24weeks achieved SVR in 100% (44/44) of treatment-naïve patients infected with HCV genotype 1, and in 91% (40/44) of patients infected with HCV genotypes 2 and 3 at 4weeks post-treatment. Addition of ribavirin had no effect on SVR rates [48]. Finally, the triple combination of daclatasvir, asunaprevir and BMS-791325, a non-nucleoside inhibitor of HCV RdRp, resulted in an SVR12 in 15 of 16 patients (94%) treated for 12weeks (data missing in the remaining patient) [49].
ABT-267
This drug candidate, developed by AbbVie, is in Phase II clinical trials. It is an oral, once-daily NS5A inhibitor that significantly reduces HCV RNA levels in vitro and in vivo. In a study of treatment-naïve genotype 1-infected patients, ABT-267 in combination with pegylated IFN-α and ribavirin produced a rapid virologic response (RVR) at 4weeks in 22 of 28 patients as compared with 2 of 22 who received placebo; after 12weeks, 25 of 28 patients receiving the NS5A inhibitor in combination with pegylated IFN-α and ribavirin showed complete early virologic response (cEVR) compared with 6 of 9 patients in the placebo group. A recently presented Phase IIb clinical trial, which used a 4-drug combination of ABT-267, ritonavir-boosted ABT-450 (a protease inhibitor), ABT-333 (a non-nucleoside inhibitor of HCV RdRp), and ribavirin achieved SVR12 in 97.5% of treatment-naïve patients and in 93.3% of prior null-responders infected with genotype 1 [50], [51]. In treatment-naïve patients, the SVR rates were 87.5% when the three drugs and ribavirin were administered for 8weeks, 89.9% when ABT-267 was administered with ABT-450 and ribavirin for 12weeks, and 87.3% when the three DAAs were administered without ribavirin for 12weeks. In null responders, the SVR rate was 88.9% with the combination of ABT-450, ABT-267, and ribavirin. Based on these results, Phase III trials with the 3 DAAs with and without ribavirin are planned [51].
Ledispasvir (GS-5885)
This oral, once-daily drug candidate, developed by Gilead Sciences, is a potent NS5A inhibitor against genotypes 1a, 1b, 4a, and 5a in vitro, but has lower activity against genotypes 2a and 3a [52]. In a randomized, placebo-controlled study of 14days of ledipasvir monotherapy in genotype 1-infected patients, significant HCV RNA reductions (up to 1000-fold) were observed. Several resistance-associated substitutions were selected, including the aforementioned Y93H and L31M. In patients infected with HCV genotype 1b, daclatasvir has been reported to be more active than ledipasvir, whereas ledipasvir has been found to be 4–5times more active than daclatasvir for the M28T and Q30H substitutions in HCV genotype 1a infection. In addition, daclatasvir has been demonstrated to be 2-fold more active against the L31M substitution as compared with ledipasvir [53]. Ledipasvir is now in a Phase II trial as a component of a 4-drug regimen with tegobuvir (a non-nucleoside inhibitor of HCV RdRp), GS-9451 (an NS3/4A protease inhibitor), and ribavirin [52]. Recent results from the ELECTRON Phase II trial have shown SVR rates 12weeks after the end of treatment of 100% in 25 treatment-naïve and 10 null responder patients infected with HCV genotype 1 with the combination of sofosbuvir, ledipasvir and ribavirin [54]. A Phase III trial with a fixed-dose combination of sofosbuvir and ledipasvir, with or without ribavirin, is in progress in treatment-naïve patients infected with HCV genotype 1 [55]. A recent presentation also showed that ledipasvir, in combination with GS-9451, pegylated IFN-α, and ribavirin achieved SVR at 4weeks post-treatment in 100% of CC IL28B patients infected with HCV genotype 1 [56].
GSK-2336805
This oral, once-daily drug candidate is being developed by GlaxoSmithKline. Preliminary studies show that GSK-2336805 is particularly effective against HCV genotype 1b, and has potent antiviral activity against other genotypes as well. A placebo-controlled Phase I study of treatment-naïve patients with chronic genotype 1 infection found a reduction in HCV RNA level of up to 1000-fold following 14days of monotherapy. This NS5A inhibitor is currently in Phase II clinical trials in treatment-naïve patients infected with HCV genotype 1 in combination with pegylated IFN-α, ribavirin, and telaprevir [57]. Resistance to GSK-2336805 maps to NS5A [58].
ACH-2928
This oral, once-daily drug candidate, developed by Achillion Pharmaceuticals, displays highly potent activity in vitro against genotype 1a replicons as well as chimeric replicons of genotypes 2–6. ACH-2928 has demonstrated in vitro synergistic activity in combination with sovaprevir (formerly ACH-1625), an HCV NS3/4A protease inhibitor, which is further enhanced by ribavirin [59]. In Phase I trials, ACH-2928 monotherapy for 3days produced up to a 3.7log10 reduction in HCV RNA levels in patients with chronic HCV genotype 1 infection [60].
BMS824393
This NS5A inhibitor is being developed by Bristol-Myers Squibb. It has shown strong in vitro potency against genotypes 1a and 1b. In a Phase I study in which this agent was used as a monotherapy for 3days in genotype 1-infected patients, a decline of up to 3.9log10 was observed [61].
IDX719
This drug candidate, developed by Idenix Pharmaceuticals, has shown greater potency in vitro than daclatasvir against HCV genotypes 1a, 1b, 2a, 3a, 4a, and 5a [62]. In Phase I studies, HCV RNA levels declined by more than 3 log10 in single-dose trials for all genotype 1, 2, and 3 patients after 24hours. Similar reductions in HCV RNA levels (over 3log10) were observed for genotype 1, 3, and 4 patients, and reductions of 2log10 for genotype 2, in 3-day monotherapy studies [63], [64]. However, evidence indicates that the Y93H substitution confers resistance to this NS5A inhibitor [62]. A Phase II clinical trial using IDX719, simeprevir (a protease inhibitor developed by Janssen and Medivir), and TMC647055, a non-nucleoside polymerase inhibitor developed by Janssen, has been announced [65].
PPI461
This oral drug candidate is under development by Presidio Pharmaceuticals. A Phase Ib trial of monotherapy for 3days in patients with HCV genotype 1 infection showed a decrease of HCV RNA level of up to 3.6log10. However, widespread resistance emerged rapidly, mapping to amino acids 28, 30, 31, and 93 [66].
PPI668
Also under development by Presidio Pharmaceuticals, this NS5A inhibitor has been shown to possess high efficacy against HCV genotype 1, with up to 3.7log10 mean HCV RNA reductions, in a Phase Ib clinical trial [67], [68]. Activity was demonstrated against variants harbouring the L31M substitution. In an added genotype-2/3 cohort, the first 2 patients achieved mean 3.0log10 RNA level reductions [68]. PPI668 will be studied in combination with two DAAs developed by Boehringer-Ingelheim, faldaprevir, an NS3/4A protease inhibitor, and BI207127, a non-nucleoside inhibitor of HCV RdRp.
ACH-3102
This NS5A inhibitor, developed by Achillion Pharmaceuticals, has a modified structure designed to have a higher pharmacologic barrier to resistance. Pharmacokinetic studies support once-daily oral dosing with this agent. ACH-3102 has shown potent antiviral activity against all genotypes in preclinical studies. In replicon studies, ACH-3102 has shown the smallest difference in potency between genotype 1a and 1b replicons, compared with daclatasvir and ACH-2928 [69]. ACH-3102 is potent against mutants harbouring substitutions that confer resistance to first-generation NS5A inhibitors (Fig. 2), such as those at positions Y93 and L31 [69]. Antiviral efficacy is also strong against double mutants that are highly resistant to other NS5A inhibitors (unpublished data). In addition, this inhibitor has shown very low potential for emergence of resistant variants in genotype 1b replicons (unpublished data). For these reasons, ACH-3102 is considered a “second-generation” NS5A inhibitor.
Fig. 2. Antiviral efficacy of ACH-3102 (second-generation NS5A inhibitor) compared with ACH-2928 and daclatasvir (first-generation NS5A inhibitors) on wild-type (parent) and mutated HCV replicons [69].

A recently reported preclinical study using ACH-3102 and ACH-2684 (an NS3/4A protease inhibitor) has shown an additive to synergistic antiviral effect against genotypes 1a and 1b without the emergence of resistance variants [70]. Recently announced results from a Phase Ia trial in patients infected with HCV genotype 1 show that a single dose of ACH-3102 produces a mean HCV RNA level reduction of up to 3.9log10, with an upper range of 4.6log10, with inhibition lasting for 4days after dosing. Moreover, ACH-3102 has a half-life of approximately 250hours (unpublished data), compared with 13–15hours for daclatasvir [71], 22–50hours for ledipasvir [53], and 25–32hours for ABT-267 [72]. A single Phase II trial has been initiated in genotype 1b patients using ACH-3102 in combination with ribavirin [73].
Progress toward all-oral combination therapies for HCV and the role of NS5A inhibitors
Currently, the standard of care for chronic HCV genotype 1 infection is a combination of pegylated IFN-α, ribavirin, and an NS3/4A protease inhibitor (i.e., boceprevir or telaprevir), whereas patients infected with other HCV genotypes continue to be treated with pegylated IFN-α and ribavirin. The SVR rates observed with the triple combination in patients infected with HCV genotype 1 range from 67% to 75% in clinical trials [74], [75]. They are probably lower in the real-life setting, indicating that a significant proportion of patients will still experience virologic failure and that improved therapeutic regimens are needed. In addition, patients receiving pegylated IFN-α and ribavirin experience a plethora of adverse effects, some of which are aggravated by the protease inhibitor [6], [7], [76]. Clinical trials of NS5A inhibitors in combination with pegylated IFN-α and ribavirin have shown promising results. However, the trials conducted thus far have included only small numbers of patients, and more studies are needed before the efficacy of such 3-drug combinations can be fully ascertained. In this respect, the results of a Phase III trial with daclatasvir, pegylated IFN-α, and ribavirin are awaited. Quadruple therapies including an NS5A inhibitor, pegylated IFN-α, ribavirin, and another DAA also appear promising. However, recent reports of very high SVR rates, over 90%, in patients treated with all-oral, IFN-free regimens with or without ribavirin clearly indicate that the IFN era is coming to an end. It is also noteworthy that NS5A inhibitors developed by one company have been used with different classes of DAAs developed by other companies [48]; as such, a highly potent NS5A inhibitor may find uses in combinations with various other DAAs to achieve high cure rates.
Due to their specificity, potency, and low EC50, NS5A inhibitors will likely be a critical component of future all-oral, IFN-free combinations. It is interesting to note that the most attractive all-oral combinations presented at the last annual meetings of the American and European liver societies all contained an NS5A inhibitor, combined either with a nucleotide analogue or a protease inhibitor and a non-nucleoside inhibitor of HCV RdRp, with or without ribavirin. Fixed-dose combinations (i.e., 2-drug combinations in 1 pill) including an NS5A inhibitor are already available in Phase II and III clinical trials. The advent of second-generation NS5A inhibitors, with a modified structure and near-equal efficacy against variants known to resist first-generation NS5A inhibitors, is also promising.

Conclusions
Although blood screening and other preventive measures have reduced the incidence of HCV in some parts of the world, infection with this virus remains a significant worldwide health concern. The multiple genotypes of HCV, as well as rapid development of mutations, have complicated the development of effective drugs. Until recently, a non-specific antiviral combination, pegylated IFN-α and ribavirin, was the mainstay of HCV therapy. The approval of two NS3/4A protease inhibitors has allowed the addition of a DAA to this treatment regimen. Although the first-generation protease inhibitors, telaprevir and boceprevir, in combination with pegylated IFN-α and ribavirin, have improved treatment of chronic HCV genotype 1 infection, response rates remain suboptimal. In addition, many patients are unable to tolerate this therapy and, among those who can, adverse events associated with the drugs can compromise patient compliance and lead to premature treatment discontinuations. Thus, there has been a strong desire to develop all-oral, IFN-free therapies with high efficacy. The discovery of the multiple roles of the NS5A protein in viral replication has been paralleled by the development of specific NS5A inhibitors. Evidence gathered thus far indicates that these agents are potent and possess antiviral activity against multiple HCV genotypes with acceptable safety profiles. In addition, clinical trial data support the efficacy of NS5A inhibitors with and without pegylated IFN-α and ribavirin, suggesting an important role for these agents as a component of all-oral therapeutic regimens for the treatment of HCV.
Financial support
Editorial assistance from ACCESS Medical was funded by Achillion Pharmaceuticals.
Conflict of interest
The author has received research grants from Gilead. He has served as an advisor for Abbott, Abbvie, Achillion, Boehringer-Ingelheim, Bristol-Myers Squibb, Gilead, Idenix, Janssen-Cilag, Madaus-Rottapharm, Merck, Novartis, and Roche.
Acknowledgments
The author would like to thank Amlan RayChaudhury, PhD, of ACCESS Medical, LLC, for editorial assistance in preparing the manuscript.
References
Source




